รายละเอียดสินค้า
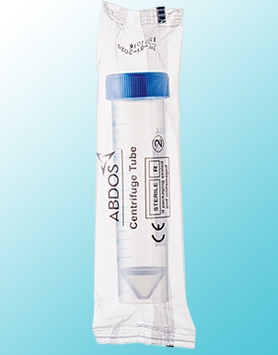
- Abdos Cryo Vials are Manufactured in state of art precision-moulded facility using high purity virgin polypropylene.
- Complies with requirement of derective 2002/72/EC & requirement of FDA regulation 21 CFR 177.1520.
- Abdos Cryo Vial is designed for long term cryogenic storage of biological sample, all type of cells and any enzymes orreagents.
- Its leak proof cap design with silicone seal and strong wall thickness makes it an ideal product for transporting samples and specimens.
- These cryo vial are designed for vapour phase liquid nitrogen freezing and also incorporate a silicone seal to enhance cap sealing.
- Manufactured to be used at temperature between -196° C to +121° C.
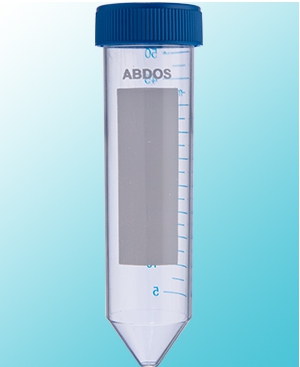
- Large white frosted portion for easy writing.
- These Cryo Vials are DNase, RNase and Pyrogen free.
- Graduation in silk-screen printing makes identification easy.
- Unique Star Foot design allows for single hand operation and grooves into Abdos P20616 single hand cryo holder rack.
- Radiation sterilized.
- Available in self-standing reclosable zipper bags.
- Best suited for IVD use (an IVD certified product)
- The silicon ring used in Abdos Cryo Vial for Sealing effects is made up of the material & ingredient under the code of federal regulation 21, as per 21 CFR 177.2600 (G) & meets USP – Class VI Regularities under plastic vial & Cap.
| Code | Capacity (ml) | Outer Diameter (mm) | Height (mm) | No. Per Case |
| P60108 | 1.0 Sterile Star Foot | 12.3 | 41.2 | 1000 |
| P60109 | 1.8 Sterile Star Foot | 12.3 | 47.5 | 1000 |
| P60110 | 1.8 Round Botom | 12.3 | 46.2 | 1000 |
| P60112 | 4.5 Sterile Star Foot | 12.3 | 90.3 | 500 |
| P60113 | 4.5 Sterile Round Foot | 12.3 | 89 | 500 |
|
*Caution: Do not use Abdos Cryogenic vial in the liquid phase of liquid nitrogen. Improper use may cause liquefied nitrogen to be trapped inside the vial and lead to pressure built up resulting in possible explosion or biohazard release. Can be used in vapour phase of liquid nitrogen. Sterile Tubes has been validated according to EN ISO 11137-2; 2007 for SAL 10-6 |
||||
ผลิตภัณฑ์แนะนำ